Dental caries
| Dental caries | |
|---|---|
| Classification and external resources | |
 Destruction of a tooth by cervical decay from dental caries. This type of decay is also known as root decay. |
|
| ICD-10 | K02. |
| ICD-9 | 521.0 |
| DiseasesDB | 29357 |
| MedlinePlus | 001055 |
Dental caries, also known as tooth decay or cavity, is a disease where bacterial processes damage hard tooth structure (enamel, dentin, and cementum).[1] These tissues progressively break down, producing dental caries (cavities, holes in the teeth). Two groups of bacteria are responsible for initiating caries: Streptococcus mutans and Lactobacillus. If left untreated, the disease can lead to pain, tooth loss, infection, and, in severe cases, death.[2] Today, caries remains one of the most common diseases throughout the world. Cariology is the study of dental caries.
The presentation of caries is highly variable; however, the risk factors and stages of development are similar. Initially, it may appear as a small chalky area that may eventually develop into a large cavitation. Sometimes caries may be directly visible, however other methods of detection such as radiographs are used for less visible areas of teeth and to judge the extent of destruction.
Tooth decay is caused by specific types of acid-producing bacteria that cause damage in the presence of fermentable carbohydrates such as sucrose, fructose, and glucose.[3][4][5] The mineral content of teeth is sensitive to increases in acidity from the production of lactic acid. Specifically, a tooth (which is primarily mineral in content) is in a constant state of back-and-forth demineralization and remineralization between the tooth and surrounding saliva. When the pH at the surface of the tooth drops below 5.5, demineralization proceeds faster than remineralization (meaning that there is a net loss of mineral structure on the tooth's surface). This results in the ensuing decay. Depending on the extent of tooth destruction, various treatments can be used to restore teeth to proper form, function, and aesthetics, but there is no known method to regenerate large amounts of tooth structure, though stem cell related research suggests one possibility. Instead, dental health organizations advocate preventive and prophylactic measures, such as regular oral hygiene and dietary modifications, to avoid dental caries.[6]
Contents |
Classification
Caries can be classified by location, etiology, rate of progression, and affected hard tissues.[7] These classification can be used to characterize a particular case of tooth decay in order to more accurately represent the condition to others and also indicate the severity of tooth destruction.
Location
Generally, there are two types of caries when separated by location: caries found on smooth surfaces and caries found in pits and fissures.[8] The location, development, and progression of smooth-surface caries differ from those of pit and fissure caries. G.V. Black created a classification system that is widely used and based on the location of the caries on the tooth. The original classification distinguished caries into five groups, indicated by the word "Class", and a Roman numeral. Pit and fissure caries is indicated as Class I; smooth surface caries is further divided into Class II, Class III, Class IV, and Class V.[9] A Class VI was added onto Black's Classification of Caries Lesions and also represents a smooth-surface carious lesion.

Pit and fissure caries (class I dental caries)
Pits and fissures are anatomic landmarks on a tooth where the enamel folds inward. Fissures are formed during the development of grooves but the enamel in the area is not fully fused. As a result, a deep linear depression forms in the enamel's surface structure, which forms a location for dental caries to develop and flourish. Fissures are mostly located on the occlusal (chewing) surfaces of posterior (rear) teeth and palatal surfaces of maxillary anterior (front) teeth. Pits are small, pinpoint depressions that are most commonly found at the ends or cross-sections of grooves.[10] In particular, buccal pits are found on the facial surfaces of molars. For all types of pits and fissures, the deep infolding of enamel makes oral hygiene along these surfaces difficult, allowing dental caries to develop more commonly in these areas.
The occlusal surfaces of teeth represent 12.5% of all tooth surfaces but are the location of over 50% of all dental caries.[11] Among children, pit and fissure caries represent 90% of all dental caries.[12] Pit and fissure caries can sometimes be difficult to detect. As the decay progresses, caries in enamel nearest the surface of the tooth spreads gradually deeper. Once the caries reaches the dentin at the dentino-enamel junction (DEJ), the decay quickly spreads laterally. Within the dentin, the decay follows a triangle pattern that points to the tooth's pulp. This pattern of decay is typically described as two triangles (one triangle in enamel, and another in dentin) with their bases conjoined to each other at the DEJ. This base-to-base pattern is typical of pit and fissure caries, unlike smooth-surface caries (where base and apex of the two triangles join).
Smooth-surface caries
There are three types of smooth-surface caries. Proximal caries, also called interproximal caries, form on the smooth surfaces between adjacent teeth. Root caries form on the root surfaces of teeth. The third type of smooth-surface caries occur on any other smooth tooth surface.

Proximal caries are the most difficult type to detect.[13] Frequently, this type of caries cannot be detected visually or manually with a dental explorer. Proximal caries form cervically (toward the roots of a tooth) just under the contact between two teeth. As a result, radiographs are needed for early discovery of proximal caries.[14] Under Black's classification system, proximal caries on posterior teeth (premolars and molars) are designated as Class II caries.[15] Proximal caries on anterior teeth (incisors and canines) are indicated as Class III if the incisal edge (chewing surface) is not included and Class IV if the incisal edge is included.
Root caries, which are sometimes described as a category of smooth-surfaces caries, are the third most common type of caries and usually occur when the root surfaces have been exposed due to gingival recession. When the gingiva is healthy, root caries is unlikely to develop because the root surfaces are not as accessible to bacterial plaque. The root surface is more vulnerable to the demineralization process than enamel because cementum begins to demineralize at 6.7 pH, which is higher than enamel's critical pH.[16] Regardless, it is easier to arrest the progression of root caries than enamel caries because roots have a greater reuptake of fluoride than enamel. Root caries are most likely to be found on facial surfaces, then interproximal surfaces, then lingual surfaces. Mandibular molars are the most common location to find root caries, followed by mandibular premolars, maxillary anteriors, maxillary posteriors, and mandibular anteriors.
Lesions on other smooth surfaces of teeth are also possible. Since these occur in all smooth surface areas of enamel except for interproximal areas, these types of caries are easily detected and are associated with high levels of plaque and diets promoting caries formation.[13] Under Black's classification system, caries near the gingiva on the facial or lingual surfaces is designated Class V.[15] Class VI is reserved for caries confined to cusp tips on posterior teeth or incisal edges of anterior teeth.
Other general descriptions
Besides the two previously mentioned categories, carious lesions can be described further by their location on a particular surface of a tooth. Caries on a tooth's surface that are nearest the cheeks or lips are called "facial caries", and caries on surfaces facing the tongue are known as "lingual caries". Facial caries can be subdivided into buccal (when found on the surfaces of posterior teeth nearest the cheeks) and labial (when found on the surfaces of anterior teeth nearest the lips). Lingual caries can also be described as palatal when found on the lingual surfaces of maxillary teeth because they are located beside the hard palate.
Caries near a tooth's cervix—the location where the crown of a tooth and its roots meet—are referred to as cervical caries. Occlusal caries are found on the chewing surfaces of posterior teeth. Incisal caries are caries found on the chewing surfaces of anterior teeth. Caries can also be described as "mesial" or "distal." Mesial signifies a location on a tooth closer to the median line of the face, which is located on a vertical axis between the eyes, down the nose, and between the contact of the central incisors. Locations on a tooth further away from the median line are described as distal.
Etiology
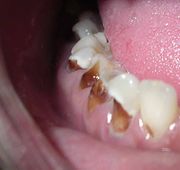
In some instances, caries are described in other ways that might indicate the cause. "Baby bottle caries", "early childhood caries", or "baby bottle tooth decay" is a pattern of decay found in young children with their deciduous (baby) teeth. The teeth most likely affected are the maxillary anterior teeth, but all teeth can be affected.[17] The name for this type of caries comes from the fact that the decay usually is a result of allowing children to fall asleep with sweetened liquids in their bottles or feeding children sweetened liquids multiple times during the day. Another pattern of decay is "rampant caries", which signifies advanced or severe decay on multiple surfaces of many teeth.[18] Rampant caries may be seen in individuals with xerostomia, poor oral hygiene, stimulant use (due to drug-induced dry mouth[19]), and/or large sugar intake. If rampant caries is a result of previous radiation to the head and neck, it may be described as radiation-induced caries. Problems can also be caused by the self destruction of roots and whole tooth resorption when new teeth erupt or later from unknown causes. Dr. Miller stated in 1887 that "Dental decay is chemico-parasitic process consisting of two stages, the decalcification of enamel, which results in its total destruction and the decalcification of dentin as a preliminary stage followed by dissolution of the softened residue." In his hypothesis, Dr.Miller assigned essential roles to three factors:
- Carbohydrate substrate.
- Acid which caused dissolution of tooth minerals.
- Oral micro organisms which produce acid and also cause proteolysis.
Rate of progression
Temporal descriptions can be applied to caries to indicate the progression rate and previous history. "Acute" signifies a quickly developing condition, whereas "chronic" describes a condition which has taken an extended time to develop where thousands of meals and snacks, many causing some acid demineralisation that is not remineralized and eventually results in cavities. Fluoride treatment can help recalcification of tooth enamel.
Recurrent caries, also described as secondary, are caries that appears at a location with a previous history of caries. This is frequently found on the margins of fillings and other dental restorations. On the other hand, incipient caries describes decay at a location that has not experienced previous decay. Arrested caries describes a lesion on a tooth which was previously demineralized but was remineralized before causing a cavitation. Using fluoride treatments can help with recalcification.
Affected hard tissue
Depending on which hard tissues are affected, it is possible to describe caries as involving enamel, dentin, or cementum. Early in its development, caries may affect only enamel. Once the extent of decay reaches the deeper layer of dentin, "dentinal caries" is used. Since cementum is the hard tissue that covers the roots of teeth, it is not often affected by decay unless the roots of teeth are exposed to the mouth. Although the term "cementum caries" may be used to describe the decay on roots of teeth, very rarely does caries affect the cementum alone. Roots have a very thin layer of cementum over a large layer of dentin, and thus most caries affecting cementum also affects dentin.
Signs and symptoms

A person experiencing caries may not be aware of the disease.[20] The earliest sign of a new carious lesion is the appearance of a chalky white spot on the surface of the tooth, indicating an area of demineralization of enamel. This is referred to as incipient decay. As the lesion continues to demineralize, it can turn brown but will eventually turn into a cavitation ("cavity"). Before the cavity forms, the process is reversible, but once a cavity forms, the lost tooth structure cannot be regenerated. A lesion which appears brown and shiny suggests dental caries were once present but the demineralization process has stopped, leaving a stain. A brown spot which is dull in appearance is probably a sign of active caries.
As the enamel and dentin are destroyed, the cavity becomes more noticeable. The affected areas of the tooth change color and become soft to the touch. Once the decay passes through enamel, the dentinal tubules, which have passages to the nerve of the tooth, become exposed and causes pain in the tooth. The pain may worsen with exposure to heat, cold, or sweet foods and drinks.[1] Dental caries can also cause bad breath and foul tastes.[21] In highly progressed cases, infection can spread from the tooth to the surrounding soft tissues. Complications such as cavernous sinus thrombosis and Ludwig's angina can be life-threatening.[22][23][24]
Causes
There are four main criteria required for caries formation: a tooth surface (enamel or dentin); caries-causing bacteria; fermentable carbohydrates (such as sucrose); and time.[25] The caries process does not have an inevitable outcome, and different individuals will be susceptible to different degrees depending on the shape of their teeth, oral hygiene habits, and the buffering capacity of their saliva. Dental caries can occur on any surface of a tooth which is exposed to the oral cavity, but not the structures which are retained within the bone.[26]
Teeth
There are certain diseases and disorders affecting teeth which may leave an individual at a greater risk for caries. Amelogenesis imperfecta, which occurs between 1 in 718 and 1 in 14,000 individuals, is a disease in which the enamel does not fully form or forms in insufficient amounts and can fall off a tooth.[27] In both cases, teeth may be left more vulnerable to decay because the enamel is not able to protect the tooth.[28]
In most people, disorders or diseases affecting teeth are not the primary cause of dental caries. Ninety-six percent of tooth enamel is composed of minerals.[29] These minerals, especially hydroxyapatite, will become soluble when exposed to acidic environments. Enamel begins to demineralize at a pH of 5.5.[30] Dentin and cementum are more susceptible to caries than enamel because they have lower mineral content.[31] Thus, when root surfaces of teeth are exposed from gingival recession or periodontal disease, caries can develop more readily. Even in a healthy oral environment, however, the tooth is susceptible to dental caries.
The anatomy of teeth may affect the likelihood of caries formation. Where the deep grooves of teeth are more numerous and exaggerated, pit and fissure caries are more likely to develop. Also, caries are more likely to develop when food is trapped between teeth.

Bacteria
The mouth contains a wide variety of oral bacteria, but only a few specific species of bacteria are believed to cause dental caries: Streptococcus mutans and Lactobacilli among them.[3][5] Lactobacillus acidophilus, Actinomyces viscosus, Nocardia spp., and Streptococcus mutans are most closely associated with caries, particularly root caries. Bacteria collect around the teeth and gums in a sticky, creamy-coloured mass called plaque, which serves as a biofilm. Some sites collect plaque more commonly than others. The grooves on the biting surfaces of molar and premolar teeth provide microscopic retention, as does the point of contact between teeth. Plaque may also collect along the gingiva.
Fermentable carbohydrates
Bacteria in a person's mouth convert glucose, fructose, and most commonly sucrose (table sugar) into acids such as lactic acid through a glycolytic process called fermentation.[4] If left in contact with the tooth, these acids may cause demineralization, which is the dissolution of its mineral content. The process is dynamic, however, as remineralization can also occur if the acid is neutralized by saliva or mouthwash. Fluoride toothpaste or dental varnish may aid remineralization.[32] If demineralization continues over time, enough mineral content may be lost so that the soft organic material left behind disintegrates, forming a cavity or hole. The impact such sugars have on the progress of dental caries is called cariogenicity. Sucrose, although a bound glucose and fructose unit, is in fact more cariogenic than a mixture of equal parts of glucose and fructose. This is due to the bacteria utilising the energy in the saccharide bond between the glucose and fructose subunits. S.mutans adheres to the biofilm on the tooth by converting sucrose into an extremely adhesive substance called dextran polysaccharide by the enzyme dextransucranase.[33]
Time
The frequency of which teeth are exposed to cariogenic (acidic) environments affects the likelihood of caries development.[34] After meals or snacks, the bacteria in the mouth metabolize sugar, resulting in an acidic by-product which decreases pH. As time progresses, the pH returns to normal due to the buffering capacity of saliva and the dissolved mineral content of tooth surfaces. During every exposure to the acidic environment, portions of the inorganic mineral content at the surface of teeth dissolves and can remain dissolved for two hours.[35] Since teeth are vulnerable during these acidic periods, the development of dental caries relies heavily on the frequency of acid exposure.
The carious process can begin within days of a tooth erupting into the mouth if the diet is sufficiently rich in suitable carbohydrates. Evidence suggests that the introduction of fluoride treatments have slowed the process.[36] Proximal caries take an average of four years to pass through enamel in permanent teeth. Because the cementum enveloping the root surface is not nearly as durable as the enamel encasing the crown, root caries tends to progress much more rapidly than decay on other surfaces. The progression and loss of mineralization on the root surface is 2.5 times faster than caries in enamel. In very severe cases where oral hygiene is very poor and where the diet is very rich in fermentable carbohydrates, caries may cause cavities within months of tooth eruption. This can occur, for example, when children continuously drink sugary drinks from baby bottles.
Other risk factors
Reduced saliva is associated with increased caries since the buffering capability of saliva is not present to counterbalance the acidic environment created by certain foods. As result, medical conditions that reduce the amount of saliva produced by salivary glands, particularly the submandibular gland and parotid gland, are likely to lead to widespread tooth decay. Examples include Sjögren's syndrome, diabetes mellitus, diabetes insipidus, and sarcoidosis.[37] Medications, such as antihistamines and antidepressants, can also impair salivary flow. Stimulants, most notoriously methylamphetamine, also occlude the flow of saliva to an extreme degree. Abusers of stimulants tend to have poor oral hygiene. Tetrahydrocannabinol, the active chemical substance in cannabis, also causes a nearly complete occlusion of salivation, known colloquially as "cotton mouth". Moreover, sixty-three percent of the most commonly prescribed medications in the United States list dry mouth as a known side effect.[37] Radiation therapy of the head and neck may also damage the cells in salivary glands, increasing the likelihood of caries formation.[38]
The use of tobacco may also increase the risk for caries formation. Some brands of smokeless tobacco contain high sugar content, increasing susceptibility to caries.[39] Tobacco use is a significant risk factor for periodontal disease, which can cause the gingiva to recede.[40] As the gingiva loses attachment to the teeth, the root surface becomes more visible in the mouth. If this occurs, root caries is a concern since the cementum covering the roots of teeth is more easily demineralized by acids than enamel.[16] Currently, there is not enough evidence to support a causal relationship between smoking and coronal caries, but evidence does suggest a relationship between smoking and root-surface caries.[41]
Intrauterine and neonatal lead exposure promote tooth decay.[42][43][44][45][46][47][48] Besides lead, all atoms with electrical charge and ionic radius similar to bivalent calcium,[49] such as cadmium, mimic the calcium ion and therefore exposure may promote tooth decay.[50]
Pathophysiology
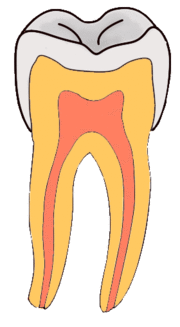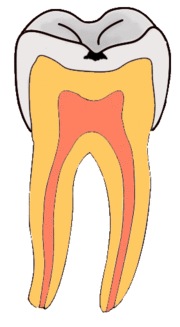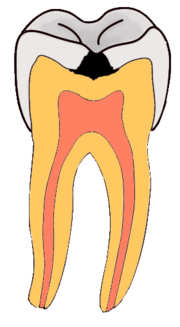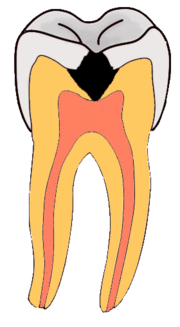
Enamel
Enamel is a highly mineralized acellular tissue, and caries act upon it through a chemical process brought on by the acidic environment produced by bacteria. As the bacteria consume the sugar and use it for their own energy, they produce lactic acid. The effects of this process include the demineralization of crystals in the enamel, caused by acids, over time until the bacteria physically penetrate the dentin. Enamel rods, which are the basic unit of the enamel structure, run perpendicularly from the surface of the tooth to the dentin. Since demineralization of enamel by caries generally follows the direction of the enamel rods, the different triangular patterns between pit and fissure and smooth-surface caries develop in the enamel because the orientation of enamel rods are different in the two areas of the tooth.[51]
As the enamel loses minerals, and dental caries progresses, the enamel develop several distinct zones, visible under a light microscope. From the deepest layer of the enamel to the enamel surface, the identified areas are the: translucent zone, dark zones, body of the lesion, and surface zone.[52] The translucent zone is the first visible sign of caries and coincides with a one to two percent loss of minerals.[53] A slight remineralization of enamel occurs in the dark zone, which serves as an example of how the development of dental caries is an active process with alternating changes.[54] The area of greatest demineralization and destruction is in the body of the lesion itself. The surface zone remains relatively mineralized and is present until the loss of tooth structure results in a cavitation.
Dentin
Unlike enamel, the dentin reacts to the progression of dental caries. After tooth formation, the ameloblasts, which produce enamel, are destroyed once enamel formation is complete and thus cannot later regenerate enamel after its destruction. On the other hand, dentin is produced continuously throughout life by odontoblasts, which reside at the border between the pulp and dentin. Since odontoblasts are present, a stimulus, such as caries, can trigger a biologic response. These defense mechanisms include the formation of sclerotic and tertiary dentin.[55]
In dentin from the deepest layer to the enamel, the distinct areas affected by caries are the translucent zone, the zone of destruction, and the zone of bacterial penetration.[51] The translucent zone represents the advancing front of the carious process and is where the initial demineralization begins. The zones of bacterial penetration and destruction are the locations of invading bacteria and ultimately the decomposition of dentin.

Sclerotic dentin
The structure of dentin is an arrangement of microscopic channels, called dentinal tubules, which radiate outward from the pulp chamber to the exterior cementum or enamel border.[56] The diameter of the dentinal tubules is largest near the pulp (about 2.5 μm) and smallest (about 900 nm) at the junction of dentin and enamel.[57] The carious process continues through the dentinal tubules, which are responsible for the triangular patterns resulting from the progression of caries deep into the tooth. The tubules also allow caries to progress faster.
In response, the fluid inside the tubules bring immunoglobulins from the immune system to fight the bacterial infection. At the same time, there is an increase of mineralization of the surrounding tubules.[58] This results in a constriction of the tubules, which is an attempt to slow the bacterial progression. In addition, as the acid from the bacteria demineralizes the hydroxyapatite crystals, calcium and phosphorus are released, allowing for the precipitation of more crystals which fall deeper into the dentinal tubule. These crystals form a barrier and slow the advancement of caries. After these protective responses, the dentin is considered sclerotic.
Fluids within dentinal tubules are believed to be the mechanism by which pain receptors are triggered within the pulp of the tooth.[59] Since sclerotic dentin prevents the passage of such fluids, pain that would otherwise serve as a warning of the invading bacteria may not develop at first. Consequently, dental caries may progress for a long period of time without any sensitivity of the tooth, allowing for greater loss of tooth structure.
Tertiary dentin
In response to dental caries, there may be production of more dentin toward the direction of the pulp. This new dentin is referred to as tertiary dentin.[57] Tertiary dentin is produced to protect the pulp for as long as possible from the advancing bacteria. As more tertiary dentin is produced, the size of the pulp decreases. This type of dentin has been subdivided according to the presence or absence of the original odontoblasts.[60] If the odontoblasts survive long enough to react to the dental caries, then the dentin produced is called "reactionary" dentin. If the odontoblasts are killed, the dentin produced is called "reparative" dentin.
In the case of reparative dentin, other cells are needed to assume the role of the destroyed odontoblasts. Growth factors, especially TGF-β,[60] are thought to initiate the production of reparative dentin by fibroblasts and mesenchymal cells of the pulp.[61] Reparative dentin is produced at an average of 1.5 μm/day, but can be increased to 3.5 μm/day. The resulting dentin contains irregularly shaped dentinal tubules which may not line up with existing dentinal tubules. This diminishes the ability for dental caries to progress within the dentinal tubules.
Diagnosis

Primary diagnosis involves inspection of all visible tooth surfaces using a good light source, dental mirror and explorer. Dental radiographs (X-rays) may show dental caries before it is otherwise visible, particularly caries between the teeth. Large dental caries are often apparent to the naked eye, but smaller lesions can be difficult to identify. Visual and tactile inspection along with radiographs are employed frequently among dentists, particularly to diagnose pit and fissure caries.[62] Early, uncavitated caries is often diagnosed by blowing air across the suspect surface, which removes moisture and changes the optical properties of the unmineralized enamel.
Some dental researchers have cautioned against the use of dental explorers to find caries.[13] In cases where a small area of tooth has begun demineralizing but has not yet cavitated, the pressure from the dental explorer could cause a cavity. Since the carious process is reversible before a cavity is present, it may be possible to arrest the caries with fluoride and remineralize the tooth surface. When a cavity is present, a restoration will be needed to replace the lost tooth structure.
At times, pit and fissure caries may be difficult to detect. Bacteria can penetrate the enamel to reach dentin, but then the outer surface may remineralize, especially if fluoride is present.[63] These caries, sometimes referred to as "hidden caries", will still be visible on x-ray radiographs, but visual examination of the tooth would show the enamel intact or minimally perforated.
Treatment

Destroyed tooth structure does not fully regenerate, although remineralization of very small carious lesions may occur if dental hygiene is kept at optimal level.[1] For the small lesions, topical fluoride is sometimes used to encourage remineralization. For larger lesions, the progression of dental caries can be stopped by treatment. The goal of treatment is to preserve tooth structures and prevent further destruction of the tooth.
Generally, early treatment is less painful and less expensive than treatment of extensive decay. Anesthetics—local, nitrous oxide ("laughing gas"), or other prescription medications—may be required in some cases to relieve pain during or following treatment or to relieve anxiety during treatment.[64] A dental handpiece ("drill") is used to remove large portions of decayed material from a tooth. A spoon is a dental instrument used to remove decay carefully and is sometimes employed when the decay in dentin reaches near the pulp.[65] Once the decay is removed, the missing tooth structure requires a dental restoration of some sort to return the tooth to functionality and aesthetic condition.
Restorative materials include dental amalgam, composite resin, porcelain, and gold.[66] Composite resin and porcelain can be made to match the color of a patient's natural teeth and are thus used more frequently when aesthetics are a concern. Composite restorations are not as strong as dental amalgam and gold; some dentists consider the latter as the only advisable restoration for posterior areas where chewing forces are great.[67] When the decay is too extensive, there may not be enough tooth structure remaining to allow a restorative material to be placed within the tooth. Thus, a crown may be needed. This restoration appears similar to a cap and is fitted over the remainder of the natural crown of the tooth. Crowns are often made of gold, porcelain, or porcelain fused to metal.

In certain cases, endodontic therapy may be necessary for the restoration of a tooth.[68] Endodontic therapy, also known as a "root canal", is recommended if the pulp in a tooth dies from infection by decay-causing bacteria or from trauma. During a root canal, the pulp of the tooth, including the nerve and vascular tissues, is removed along with decayed portions of the tooth. The canals are instrumented with endodontic files to clean and shape them, and they are then usually filled with a rubber-like material called gutta percha.[69] The tooth is filled and a crown can be placed. Upon completion of a root canal, the tooth is now non-vital, as it is devoid of any living tissue.
An extraction can also serve as treatment for dental caries. The removal of the decayed tooth is performed if the tooth is too far destroyed from the decay process to effectively restore the tooth. Extractions are sometimes considered if the tooth lacks an opposing tooth or will probably cause further problems in the future, as may be the case for wisdom teeth.[70] Extractions may also be preferred by patients unable or unwilling to undergo the expense or difficulties in restoring the tooth.
Medicinal plants in the treatment of dental caries
| S. No | Botanical Name | Part used | Inhibition Organisms |
|---|---|---|---|
| 1. | Acacia leucophloea | Bark | Streptococcus mutans |
| 2. | Albizia lebbeck | Bark | Streptococcus mutans |
| 3. | Abies canadensis | Whole plant | Streptococcus mutans |
| 4. | Aristolochia cymbifera | Whole plant | Streptococcus mutans |
| 5. | Annona senegalensis | Whole plant | Streptococcus mutans |
| 6. | Albizia julibrissin | Whole plant | Streptococcus mutans |
| 7. | Allium sativum | Bulbs | Streptococcus mutans |
| 8. | Anacyclus pyrethrum | Root | Streptococcus mutans |
| 9. | Areca catechu | Nuts | Streptococcus mutans |
| 10. | Breynia nivosus | Whole plant | Streptococcus mutans |
| 14. | Citrus medica | Roots | Streptococcus mutans |
| 15. | Coptidis rhizoma | Whole plant | Streptococcus mutans |
| 16. | Caesalpinia martius | Fruits | Streptococcus mutans, Streptococcus oralis, Lactobacillus casei |
| 17. | Cocos nucifera | Whole plant | Streptococcus mutans |
| 18. | Caesalpinia pyramidalis | Whole plant | Streptococcus mutans |
| 19. | Chelidonium majus | Whole plant | Streptococcus mutans |
| 20. | Drosera peltata | Whole plant | Streptococcus mutans, Streptococcus sobrinus |
| 21. | Embelia ribes | Fruit | Streptococcus mutans |
| 22. | Erythrina variegata | Root | Streptococcus mutans, Streptococcus sanguis |
| 23. | Euclea natalensis | Whole plant | Streptococcus mutans |
| 24. | Fiscus microcarpa | Aerial part | Streptococcus mutans |
| 25. | Gymnema Sylvester | Leaves,Roots | Streptococcus mutans |
| 27. | Glycyrrhiza glabra | Root | Streptococcus mutans |
| 28. | Hamamelis virginiana | Leaves | Preveotella spp., Actinomyces odontolitycus |
| 29. | Harungana madagascariensis | Leaves | Actinomyces, Fusobacterium, Lactobacillus, Prevotella, Propioni bacterium, Streptococcus spp. |
| 30. | Helichrysum italicum | Whole plant | Streptococcus mutans, Streptococcus sanguis, Streptococcus sobrinus |
| 31. | Ginkgo biloba | Whole plant | Streptococcus mutans |
| 32. | Juniperus virginiana | Whole plant | Streptococcus mutans |
| 33. | Kaemperia pandurata | Dried rhizomes, root | Streptococcus mutans |
| 34. | Legenaria sicerania | Leaves | Streptococcus mutans |
| 35. | Mentha arvensis | Leaves | Streptococcus mutans |
| 36. | Mikania lavigata | Aerial parts | Streptococcus mutans, Streptococcus sobrinus |
| 37. | Mikania glomerata | Whole plant | Streptococcus cricetus |
| 38. | Melissa officinalis | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 39. | Magnolia grandiflora | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 40. | Melissa officinalis | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 41. | Magnolia grandiflora | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 42. | Nicotiana tabacum | leaves | Streptococcus mutans |
| 43. | Physalis angulata | Flower | Streptococcus mutans |
| 44. | Pinus virginiana | Whole plant | Streptococcus mutans |
| 45. | Pistacia lentiscus | mastic gum | Porphyromonas gingivalis |
| 46. | Pistacia vera | Whole plant | oral Streptococci |
| 47. | Piper cubeba | Whole plant | periodontal pathogens |
| 48. | Polygonum cuspidatum | Root | Streptococcus mutans, Streptococcus sobrinus |
| 49. | Rheedia brasiliensis | Fruit | Streptococcus mutans |
| 50. | Rhus corriaria | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 51. | Rhus corriaria | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 52. | Rosmarinus officinalis | Whole plant | Streptococcus mutans |
| 53. | Quercus infectoria | Gall | Streptococcus mutans |
| 54. | Rhus corriaria | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 55. | Syzygium cumini | Bark | Streptococcus mutans |
| 56. | Sassafras albidum | Whole plant | Streptococcus mutans |
| 57. | Solanum xathaocarpum | Whole plant | Streptococcus mutans |
| 58. | Syzygium aromaticum | Dried flower | Staphylococcus aureus |
| 59. | Thymus vulgaris | Whole plant | Streptococcus mutans, Streptococcus sanguis |
| 60. | Tanacetum vulgare | Whole plant | Staphylococcus aureus |
| 61. | Thuja plicata | Whole plant | Staphylococcus aureus |
| 62. | Ziziphus joazeiro | Whole plant | Staphylococcus aureus |
Prevention

Oral hygiene
Personal hygiene care consists of proper brushing and flossing daily.[6] The purpose of oral hygiene is to minimize any etiologic agents of disease in the mouth. The primary focus of brushing and flossing is to remove and prevent the formation of plaque. Plaque consists mostly of bacteria.[72] As the amount of bacterial plaque increases, the tooth is more vulnerable to dental caries when carbohydrates in the food are left on teeth after every meal or snack. A toothbrush can be used to remove plaque on accessible surfaces, but not between teeth or inside pits and fissures on chewing surfaces. When used correctly, dental floss removes plaque from areas which could otherwise develop proximal caries. Other adjunct hygiene aids include interdental brushes, water picks, and mouthwashes.
However oral hygiene is probably more effective at preventing gum disease than tooth decay. The brush and fluoride toothpaste have no access inside pits and fissures, where chewing forces food to be trapped. (Occlusal caries accounts for between 80 and 90 percent of caries in children (Weintraub, 2001). The teeth at highest risk for carious lesions are the first and second permanent molars.)
Professional hygiene care consists of regular dental examinations and cleanings. Sometimes, complete plaque removal is difficult, and a dentist or dental hygienist may be needed. Along with oral hygiene, radiographs may be taken at dental visits to detect possible dental caries development in high risk areas of the mouth.
Dietary modification
For dental health, frequency of sugar intake is more important than the amount of sugar consumed.[34] In the presence of sugar and other carbohydrates, bacteria in the mouth produce acids which can demineralize enamel, dentin, and cementum. The more frequently teeth are exposed to this environment, the more likely dental caries are to occur. Therefore, minimizing snacking is recommended, since snacking creates a continual supply of nutrition for acid-creating bacteria in the mouth. Also, chewy and sticky foods (such as dried fruit or candy) tend to adhere to teeth longer, and consequently are best eaten as part of a meal. Brushing the teeth after meals is recommended. For children, the American Dental Association and the European Academy of Paediatric Dentistry recommend limiting the frequency of consumption of drinks with sugar, and not giving baby bottles to infants during sleep.[73][74] Mothers are also recommended to avoid sharing utensils and cups with their infants to prevent transferring bacteria from the mother's mouth.[75]
It has been found that milk and certain kinds of cheese like Cheddar can help counter tooth decay if eaten soon after the consumption of foods potentially harmful to teeth.[34] Also, chewing gum containing xylitol (a sugar alcohol) is widely used to protect teeth in some countries, being especially popular in the Finnish candy industry.[76] Xylitol's effect on reducing plaque is probably due to bacteria's inability to utilize it like other sugars.[77] Chewing and stimulation of flavour receptors on the tongue are also known to increase the production and release of saliva, which contains natural buffers to prevent the lowering of pH in the mouth to the point where enamel may become demineralised.[78]

Other preventive measures
The use of dental sealants is a means of prevention. A sealant is a thin plastic-like coating applied to the chewing surfaces of the molars. This coating prevents food being trapped inside pits and fissures in grooves under chewing pressure so resident plaque bacteria are deprived of carbohydrate that they change to acid demineralisation and thus prevents the formation of pit and fissure caries, the most common form of dental caries. Sealants are usually applied on the teeth of children, shortly after the molars erupt. Older people may also benefit from the use of tooth sealants, but their dental history and likelihood of caries formation are usually taken into consideration.
Calcium, as found in food such as milk and green vegetables, are often recommended to protect against dental caries. It has been demonstrated that calcium and fluoride supplements decrease the incidence of dental caries. Fluoride helps prevent decay of a tooth by binding to the hydroxyapatite crystals in enamel.[79] The incorporated calcium makes enamel more resistant to demineralization and, thus, resistant to decay.[80] Topical fluoride is also recommended to protect the surface of the teeth. This may include a fluoride toothpaste or mouthwash. Many dentists include application of topical fluoride solutions as part of routine visits.
Other products with little or less supportive scientific evidence for effectiveness for the purpose of remineralization include DCPD, ACP, calcium compounds, fluoride, and Enamelon.
Remineralization can also be performed professionally at the dentist.
Furthermore, recent research shows that low intensity laser radiation of argon ion lasers may prevent the susceptibility for enamel caries and white spot lesions.[81]
As bacteria are a major factor contributing to poor oral health, there is currently research to find a vaccine for dental caries. As of 2004, such a vaccine has been successfully tested on animals,[82] and is in clinical trials for humans as of May 2006.[83]
Chewing gum after eating promotes the flow of saliva which naturally reduces the acidic pH environment and promotes remineralization.
Xylitol lollies and gum also inhibit the growth of Streptococcus mutans.
Epidemiology

Worldwide, most children and an estimated ninety percent of adults have experienced caries, with the disease most prevalent in Asian and Latin American countries and least prevalent in African countries.[85] In the United States, dental caries is the most common chronic childhood disease, being at least five times more common than asthma.[86] It is the primary pathological cause of tooth loss in children.[87] Between twenty-nine and fifty-nine percent of adults over the age of fifty experience caries.[88]
The number of cases has decreased in some developed countries, and this decline is usually attributed to increasingly better oral hygiene practices and preventive measures such as fluoride treatment.[89] Nonetheless, countries that have experienced an overall decrease in cases of tooth decay continue to have a disparity in the distribution of the disease.[88] Among children in the United States and Europe, twenty percent of the population endures sixty to eighty percent of cases of dental caries.[90] A similarly skewed distribution of the disease is found throughout the world with some children having none or very few caries and others having a high number.[88] Australia, Nepal, and Sweden have a low incidence of cases of dental caries among children, whereas cases are more numerous in Costa Rica and Slovakia.[91]
The classic "DMF" (decay/missing/filled) index is one of the most common methods for assessing caries prevalence as well as dental treatment needs among populations. This index is based on in-field clinical examination of individuals by using a probe, mirror and cotton rolls. Because the DMF index is done without X-ray imaging, it underestimates real caries prevalence and treatment needs.[63]
History

There is a long history of dental caries. Over a million years ago, hominids such as Australopithecus suffered from cavities.[92] The largest increases in the prevalence of caries have been associated with dietary changes.[92][93] Archaeological evidence shows that tooth decay is an ancient disease dating far into prehistory. Skulls dating from a million years ago through the neolithic period show signs of caries, excepting those from the Paleolithic and Mesolithic ages.[92] The increase of caries during the neolithic period may be attributed to the increased consumption of plant foods containing carbohydrates.[94] The beginning of rice cultivation in South Asia is also believed to have caused an increase in caries.
A Sumerian text from 5000 BC describes a "tooth worm" as the cause of caries.[95] Evidence of this belief has also been found in India, Egypt, Japan, and China.[93] Unearthed ancient skulls show evidence of primitive dental work. In Pakistan, teeth dating from around 5500 BC to 7000 BC show nearly perfect holes from primitive dental drills.[96] The Ebers Papyrus, an Egyptian text from 1550 BC, mentions diseases of teeth.[95] During the Sargonid dynasty of Assyria during 668 to 626 BC, writings from the king's physician specify the need to extract a tooth due to spreading inflammation.[93] In the Roman Empire, wider consumption of cooked foods led to a small increase in caries prevalence.[90] The Greco-Roman civilization, in addition to the Egyptian, had treatments for pain resulting from caries.[93]
The rate of caries remained low through the Bronze Age and Iron Age, but sharply increased during the Middle Ages.[92] Periodic increases in caries prevalence had been small in comparison to the 1000 AD increase, when sugar cane became more accessible to the Western world. Treatment consisted mainly of herbal remedies and charms, but sometimes also included bloodletting.[97] The barber surgeons of the time provided services that included tooth extractions.[93] Learning their training from apprenticeships, these health providers were quite successful in ending tooth pain and likely prevented systemic spread of infections in many cases. Among Roman Catholics, prayers to Saint Apollonia, the patroness of dentistry, were meant to heal pain derived from tooth infection.[98]
There is also evidence of caries increase in North American Indians after contact with colonizing Europeans. Before colonization, North American Indians subsisted on hunter-gatherer diets, but afterwards there was a greater reliance on maize agriculture, which made these groups more susceptible to caries.[92]
In the medieval Islamic world, Muslim physicians such as al-Gazzar and Avicenna (in The Canon of Medicine) provided the earliest known treatments for caries, though they also believed that it was caused by tooth worms as the ancients had. This was eventually proven false in 1200 by another Muslim dentist named Gaubari, who in his Book of the Elite concerning the unmasking of mysteries and tearing of veils, was the first to reject the idea of caries being caused by tooth worms, and he stated that tooth worms in fact do not even exist. The theory of the tooth worm was thus no longer accepted in the Islamic medical community from the 13th century onwards.[99]
During the European Age of Enlightenment, the belief that a "tooth worm" caused caries was also no longer accepted in the European medical community.[100] Pierre Fauchard, known as the father of modern dentistry, was one of the first to reject the idea that worms caused tooth decay and noted that sugar was detrimental to the teeth and gingiva.[101] In 1850, another sharp increase in the prevalence of caries occurred and is believed to be a result of widespread diet changes.[93] Prior to this time, cervical caries was the most frequent type of caries, but increased availability of sugar cane, refined flour, bread, and sweetened tea corresponded with a greater number of pit and fissure caries.
In the 1890s, W.D. Miller conducted a series of studies that led him to propose an explanation for dental caries that was influential for current theories. He found that bacteria inhabited the mouth and that they produced acids which dissolved tooth structures when in the presence of fermentable carbohydrates.[102] This explanation is known as the chemoparasitic caries theory.[103] Miller's contribution, along with the research on plaque by G.V. Black and J.L. Williams, served as the foundation for the current explanation of the etiology of caries.[93] Several of the specific strains of bacteria were identified in 1921 by Fernando E. Rodriguez Vargas.
See also
- Feline odontoclastic resorptive lesion
- Acid erosion
- Oral microbiology
Footnotes and sources
- ↑ 1.0 1.1 1.2 MedlinePlus Encyclopedia Dental Cavities
- ↑ Cavities/tooth decay, hosted on the Mayo Clinic website. Page accessed May 25, 2008.
- ↑ 3.0 3.1 Hardie JM (May 1982). "The microbiology of dental caries". Dent Update 9 (4): 199–200, 202–4, 206–8. PMID 6959931.
- ↑ 4.0 4.1 Holloway PJ; Moore, W.J. (September 1983). "The role of sugar in the etiology of dental caries". J Dent 11 (3): 189–213. doi:10.1016/0300-5712(83)90182-3.
Moore WJ; Moore, W.J. (September 1983). "1. Sugar and the antiquity of dental caries". J Dent 11 (3): 189–90. doi:10.1016/0300-5712(83)90182-3. PMID 6358295.
Rugg-Gunn AJ, Murray JJ (September 1983). "2. The epidemiological evidence". J Dent 11 (3): 190–9. doi:10.1016/0300-5712(83)90183-5. PMID 6358296.
Edgar WM (September 1983). "3. The physiochemical evidence". J Dent 11 (3): 199–205. doi:10.1016/0300-5712(83)90184-7. PMID 6358297.
Drucker DB (September 1983). "4. The microbiological evidence". J Dent 11 (3): 205–7. doi:10.1016/0300-5712(83)90185-9. PMID 6358298.
Ryan LA (September 1983). "5. Confectionery and dental caries". J Dent 11 (3): 207–9. doi:10.1016/0300-5712(83)90186-0. PMID 6358299.
Shaw JH (September 1983). "6. Evidence from experimental animal research". J Dent 11 (3): 209–13. doi:10.1016/0300-5712(83)90187-2. PMID 6417207. - ↑ 5.0 5.1 Rogers AH (editor). (2008). Molecular Oral Microbiology. Caister Academic Press. ISBN 978-1-904455-24-0. http://www.horizonpress.com/oral2.
- ↑ 6.0 6.1 Oral Health Topics: Cleaning your teeth and gums. Hosted on the American Dental Association website. Page accessed August 15, 2006.
- ↑ Sonis, Stephen T. (2003). Dental Secrets (3rd ed.). Philadelphia: Hanley & Belfus. pp. 130. ISBN 1-56053-573-3.
- ↑ Schwartz RB,.; Summitt, James B.; Robbins, J. William (2001). Fundamentals of operative dentistry: a contemporary approach (2nd ed.). Chicago: Quintessence Pub. Co. pp. 30. ISBN 0-86715-382-2.
- ↑ Scheid, Rickne C. "Woelfel's Dental Anatomy: Its Relevance to Dentistry." 7th edition. Lippincott Williams & Wilkins, 2007, p. 434. ISBN 0-78176-860-8. Preview located here.
- ↑ Ash & Nelson, "Wheeler's Dental Anatomy, Physiology, and Occlusion." 8th edition. Saunders, 2003, p. 13. ISBN 0-7216-9382-2.
- ↑ Doniger, Sheri, B. "Sealed." Dental Economics, 2003. Page accessed August 13, 2006.
- ↑ Oral Health Resources - Dental Caries Fact Sheet. Hosted on the Centers for Disease Control and Prevention website. Page accessed August 13, 2006.
- ↑ 13.0 13.1 13.2 Summit, James B., J. William Robbins, and Richard S. Schwartz. "Fundamentals of Operative Dentistry: A Contemporary Approach." 2nd edition. Carol Stream, Illinois, Quintessence Publishing Co, Inc, 2001, p. 31. ISBN 0-86715-382-2.
- ↑ Health Strategy Oral Health Toolkit, hosted by the New Zealand's Ministry of Health. Page accessed August 15, 2006.
- ↑ 15.0 15.1 Qualtrough, A. J. E. , J D Satterthwaite, L A Morrow, Paul A. Brunton. "Principles of Operative Dentistry." Blackwell Publishing, 2005, p. 28. ISBN 1-40511-821-0.
- ↑ 16.0 16.1 Banting, D.W. "The Diagnosis of Root Caries." Presentation to the National Institute of Health Consensus Development Conference on Diagnosis and Management of Dental Caries Throughout Life, in pdf format, hosted on the National Institute of Dental and Craniofacial Research, p. 19. Page accessed August 15, 2006.
- ↑ ADA Early Childhood Tooth Decay (Baby Bottle Tooth Decay). Hosted on the American Dental Association website. Page accessed August 14, 2006.
- ↑ Radiographic Classification of Caries. Hosted on the Ohio State University website. Page accessed August 14, 2006.
- ↑ ADA Methamphetamine Use (METH MOUTH). Hosted on the American Dental Association website. Page accessed February 14, 2007.
- ↑ Health Promotion Board: Dental Caries, affiliated with the Singapore government. Page accessed August 14, 2006.
- ↑ Tooth Decay, hosted on the New York University Medical Center website. Page accessed August 14, 2006.
- ↑ Cavernous Sinus Thrombosis, hosted on WebMD. Page accessed May 25, 2008.
- ↑ MedlinePlus Encyclopedia Ludwig's Anigna
- ↑ Hartmann, Richard W. Ludwig's Angina in Children, hosted on the American Academy of Family Physicians website. Page accessed May 25, 2008.
- ↑ Southam JC, Soames JV (1993). "2. Dental Caries". Oral pathology (2nd ed.). Oxford: Oxford Univ. Press. ISBN 0-19-262214-5.
- ↑ Smith B, Pickard HM, Kidd EAM (1990). "1. Why restore teeth?". Pickard's manual of operative dentistry (6th ed.). Oxford [Oxfordshire]: Oxford University Press. ISBN 0-19-261808-3.
- ↑ Neville, B.W., Douglas Damm, Carl Allen, Jerry Bouquot. "Oral & Maxillofacial Pathology." 2nd edition, 2002, p. 89. ISBN 0-7216-9003-3.
- ↑ Neville, B.W., Douglas Damm, Carl Allen, Jerry Bouquot. "Oral & Maxillofacial Pathology." 2nd edition, 2002, p. 94. ISBN 0-7216-9003-3.
- ↑ Cate, A.R. Ten. "Oral Histology: development, structure, and function." 5th edition, 1998, p. 1. ISBN 0-8151-2952-1.
- ↑ Dawes C (December 2003). "What is the critical pH and why does a tooth dissolve in acid?". J Can Dent Assoc 69 (11): 722–4. PMID 14653937. http://www.cda-adc.ca/jcda/vol-69/issue-11/722.html.
- ↑ Mellberg JR (1986). "Demineralization and remineralization of root surface caries". Gerodontology 5 (1): 25–31. doi:10.1111/j.1741-2358.1986.tb00380.x. PMID 3549537.
- ↑ Silverstone LM (May 1983). "Remineralization and enamel caries: new concepts". Dent Update 10 (4): 261–73. PMID 6578983.
- ↑ Madigan M.T. & Martinko J.M. Brock - Biology of Microorganisms. 11th Ed. Pearson, USA. pp. 705
- ↑ 34.0 34.1 34.2 "Dental Health", hosted on the British Nutrition Foundation website, 2004. Page accessed August 13, 2006.
- ↑ Dental Caries, hosted on the University of California Los Angeles School of Dentistry website. Page accessed August 14, 2006.
- ↑ Summit, James B., J. William Robbins, and Richard S. Schwartz. "Fundamentals of Operative Dentistry: A Contemporary Approach." 2nd edition. Carol Stream, Illinois, Quintessence Publishing Co, Inc, 2001, p. 75. ISBN 0-86715-382-2.
- ↑ 37.0 37.1 Neville, B.W., Douglas Damm, Carl Allen, Jerry Bouquot. "Oral & Maxillofacial Pathology." 2nd edition, 2002, p. 398. ISBN 0-7216-9003-3.
- ↑ Oral Complications of Chemotherapy and Head/Neck Radiation, hosted on the National Cancer Institute website. Page accessed January 8, 2007.
- ↑ Neville, B.W., Douglas Damm, Carl Allen, Jerry Bouquot. "Oral & Maxillofacial Pathology." 2nd edition, 2002, p. 347. ISBN 0-7216-9003-3.
- ↑ Tobacco Use Increases the Risk of Gum Disease, hosted on the American Academy of Periodontology. Page accessed January 9, 2007.
- ↑ Executive Summary of U.S. Surgeon General's report titled, "The Health Consequences of Smoking: A Report of the Surgeon General," hosted on the CDC website, p. 12. Page accessed January 9, 2007.
- ↑ Brudevold F, Steadman LT (1956). "The distribution of lead in human enamel". J Dent Res 35 (3): 430–437. PMID 13332147. http://jdr.sagepub.com/cgi/reprint/35/3/430.pdf.
- ↑ Brudevold F, Aasenden R, Srinivasian BN, Bakhos Y (1977). "Lead in enamel and saliva, dental caries and the use of enamel biopsies for measuring past exposure to lead.". J Dent Res 56 (10): 1165–1171. PMID 272374. http://jdr.sagepub.com/cgi/reprint/56/10/1165.pdf.
- ↑ Goyer RA (1990). "Transplacental transport of lead". Environ Health Perspect (Brogan &) 89: 101–105. doi:10.2307/3430905. PMID 2088735. PMC 1567784. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1567784/pdf/envhper00422-0101.pdf.
- ↑ Moss ME, Lamphear BP, Auinger P (1999). "Association of dental caries and blood lead levels". JAMA 281 (24): 2294–2298. doi:10.1001/jama.281.24.2294. PMID 10386553. http://jama.ama-assn.org/cgi/content/full/281/24/2294.
- ↑ Campbell JR, Moss ME, Raubertas RF (2000). "The association between caries and childhood lead exposure". Environ Health Perspect (Brogan &) 108 (11): 1099–1102. doi:10.2307/3434965. PMID 11102303. PMC 1240169. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1240169/pdf/ehp0108-001099.pdf.
- ↑ Gemmel A, Tavares M, Alperin S, Soncini J, Daniel D, Dunn J,Crawford S, Braveman N, Clarkson TW, McKinlay S, Bellinger DC (2002). "Blood Lead Level and Dental Caries in School-Age Children". Environ Health Perspect 110 (10): A625–A630. PMID 12361944. PMC 1241049. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1241049/pdf/ehp0110-a00625.pdf.
- ↑ Billings RJ, Berkowitz RJ, Watson G (2004). "Teeth". Pediatrics 113 (4): 1120–1127. PMID 15060208. http://pediatrics.aappublications.org/cgi/reprint/113/4/S1/1120.pdf.
- ↑ Leroy N, Bres E. (2001). "Structure and substitutions in fluorapatite.". Eur Cell Mater. 2: 36–48. PMID 14562256. http://www.ecmjournal.org/journal/papers/vol002/pdf/v002a05.pdf.
- ↑ Arora M, Weuve J, Schwartz J, Wright RO (2008). "Association of environmental cadmium exposure with pediatric dental caries". Environ Health Perspect. 116 (6): 821–825. doi:10.1289/ehp.10947. PMID 8909881. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2430240/pdf/ehp0116-000821.pdf.
- ↑ 51.0 51.1 Kidd EA, Fejerskov O (1 July 2004). "What constitutes dental caries? Histopathology of carious enamel and dentin related to the action of cariogenic biofilms". J Dent Res. 83 (Spec No C): C35–8. doi:10.1177/154405910408301S07. PMID 15286119. http://jdr.iadrjournals.org/cgi/pmidlookup?view=long&pmid=15286119.
- ↑ Darling AI (1963). "Resistance of the Enamel to Dental Caries". J Dent Res. 42 (1): 488–96. PMID 14041429. http://jdr.iadrjournals.org/cgi/reprint/42/1/488.pdf.
- ↑ Robinson C, Shore RC, Brookes SJ, Strafford S, Wood SR, Kirkham J (2000). "The chemistry of enamel caries". Crit Rev Oral Biol Med. 11 (4): 481–95. doi:10.1177/10454411000110040601. PMID 11132767. http://crobm.iadrjournals.org/cgi/reprint/11/4/481.pdf.
- ↑ Cate, A.R. Ten. "Oral Histology: development, structure, and function." 5th edition, 1998, p. 417. ISBN 0-8151-2952-1.
- ↑ "Teeth & Jaws: Caries, Pulp, & Periapical Conditions," hosted on the University of Southern California School of Dentistry website. Page accessed June 22, 2007.
- ↑ Ross, Michael H., Gordon I. Kaye, and Wojciech Pawlina, 2003. Histology: a text and atlas. 4th edition, p. 450. ISBN 0-683-30242-6.
- ↑ 57.0 57.1 Cate, A.R. Ten. "Oral Histology: development, structure, and function." 5th edition, 1998, p. 152. ISBN 0-8151-2952-1.
- ↑ Summit, James B., J. William Robbins, and Richard S. Schwartz. "Fundamentals of Operative Dentistry: A Contemporary Approach." 2nd edition. Carol Stream, Illinois, Quintessence Publishing Co, Inc, 2001, p. 13. ISBN 0-86715-382-2.
- ↑ Dababneh RH, Khouri AT, Addy M (December 1999). "Dentine hypersensitivity - an enigma? A review of terminology, mechanisms, aetiology and management". Br Dent J 187 (11): 606–11; discussion 603. doi:10.1038/sj.bdj.4800345a. PMID 16163281. http://www.nature.com/bdj/journal/v187/n11/full/4800345a.html.
The referred to theory is the widely accepted hydrodynamic theory of sensitivity. - ↑ 60.0 60.1 Smith AJ, Murray PE, Sloan AJ, et al. (August 2001). "Trans-dentinal Stimulation of Tertiary Dentinogenesis". Advances in Dental Research 15 (1): 51–4. doi:10.1177/08959374010150011301. PMID 12640740. http://adr.iadrjournals.org/cgi/reprint/15/1/51.pdf.
- ↑ Summit, James B., J. William Robbins, and Richard S. Schwartz. "Fundamentals of Operative Dentistry: A Contemporary Approach." 2nd edition. Carol Stream, Illinois, Quintessence Publishing Co, Inc, 2001, p. 14. ISBN 0-86715-382-2.
- ↑ Rosenstiel, Stephen F. Clinical Diagnosis of Dental Caries: A North American Perspective. Maintained by the University of Michigan Dentistry Library, along with the National Institutes of Health, National Institute of Dental and Craniofacial Research. 2000. Page accessed August 13, 2006.
- ↑ 63.0 63.1 Zadik Yehuda, Bechor Ron (June/July 2008). "Hidden Occlusal Caries - Challenge for the Dentist" (PDF). New York State Dental Journal 74 (4): 46–50. PMID 18788181. http://www.nysdental.org/img/current-pdf/JrnlJuneJuly2008.pdf. Retrieved 2008-08-08.
- ↑ Oral Health Topics: Anesthesia Frequently Asked Questions, hosted on the American Dental Association website. Page accessed August 16, 2006.
- ↑ Summit, James B., J. William Robbins, and Richard S. Schwartz. "Fundamentals of Operative Dentistry: A Contemporary Approach." 2nd edition. Carol Stream, Illinois, Quintessence Publishing Co, Inc, 2001, p. 128. ISBN 0-86715-382-2.
- ↑ "Aspects of Treatment of Cavities and of Caries Disease" from the Disease Control Priorities Project. Page accessed August 15, 2006.
- ↑ Oral Health Topics: Dental Filling Options, hosted on the American Dental Association website. Page accessed August 16, 2006.
- ↑ What is a Root Canal?, hosted by the Academy of General Dentistry. Page accessed August 16, 2006.
- ↑ FAQs About Root Canal Treatment, hosted on the American Association of Endodontists website. Page accessed August 16, 2006.
- ↑ Wisdom Teeth, packet in pdf format hosted by the American Association of Oral and Maxillofacial Surgeons. Page accessed August 16, 2006.
- ↑ B.Parimala Devi et al. / Journal of Pharmacy Research 2009, 2(11),1669-1675 http://jpronline.info/article/view/906/708
- ↑ Introduction to Dental Plaque. Hosted on the Leeds Dental Institute Website. Page accessed August 14, 2006.
- ↑ A Guide to Oral Health to Prospective Mothers and their Infants, hosted on the European Academy of Paediatric Dentistry website. Page accessed August 14, 2006.
- ↑ Oral Health Topics: Baby Bottle Tooth Decay, hosted on the American Dental Association website. Page accessed August 14, 2006.
- ↑ Guideline on Infant Oral Health Care, hosted on the American Academy of Pediatric Dentistry website. Page accessed January 13, 2007.
- ↑ "History", hosted on the Xylitol.net website. Page accessed October 22, 2006.
- ↑ Ly KA, Milgrom P, Roberts MC, Yamaguchi DK, Rothen M, Mueller G (2006). "Linear response of mutans streptococci to increasing frequency of xylitol chewing gum use: a randomized controlled trial [ISRCTN43479664"]. BMC Oral Health 6: 6. doi:10.1186/1472-6831-6-6. PMID 16556326.
- ↑ Bots CP, Brand HS, Veerman EC, van Amerongen BM, Nieuw Amerongen AV (June 2004). "Preferences and saliva stimulation of eight different chewing gums". Int Dent J 54 (3): 143–8. PMID 15218894.
- ↑ Cate, A.R. Ten. "Oral Histology: development, structure, and function." 5th edition, 1998, p. 223. ISBN 0-8151-2952-1.
- ↑ Ross, Michael H., Gordon I. Kaye, and Wojciech Pawlina, 2003. "Histology: a text and atlas." 4th edition, p. 453. ISBN 0-683-30242-6.
- ↑ Westerman GH, Hicks MJ, Flaitz CM, Powell GL (1 May 2006). "In vitro caries formation in primary tooth enamel: role of argon laser irradiation and remineralizing solution treatment". J Am Dent Assoc 137 (5): 638–44. PMID 16739544. http://jada.ada.org/cgi/pmidlookup?view=long&pmid=16739544.
- ↑ New Drill for Tomorrow's Dentists. WIRED Magazine, May, 2004. Page accessed May 24, 2007.
- ↑ "Planet Biotechnology:Products". Planet Biotechnology. http://www.planetbiotechnology.com/products.html.
- ↑ "WHO Disease and injury country estimates". World Health Organization. 2009. http://www.who.int/healthinfo/global_burden_disease/estimates_country/en/index.html. Retrieved Nov. 11, 2009.
- ↑ The World Oral Health Report 2003: Continuous improvement of oral health in the 21st century – the approach of the WHO Global Oral Health Programme, released by the World Health Organization. (File in pdf format.) Page accessed August 15, 2006.
- ↑ Healthy People: 2010. Html version hosted on Healthy People.gov website. Page accessed August 13, 2006.
- ↑ Frequently Asked Questions, hosted on the American Dental Hygiene Association website. Page accessed August 15, 2006.
- ↑ 88.0 88.1 88.2 "Dental caries", from the Disease Control Priorities Project. Page accessed August 15, 2006.
- ↑ World Health Organization website, "World Water Day 2001: Oral health", p. 2. Page accessed August 14, 2006.
- ↑ 90.0 90.1 Touger-Decker R, van Loveren C (1 October 2003). "Sugars and dental caries". Am J Clin Nutr. 78 (4): 881S–92S. PMID 14522753. http://www.ajcn.org/cgi/pmidlookup?view=long&pmid=14522753.
- ↑ "Table 38.1. Mean DMFT and SiC Index of 12-Year-Olds for Some Countries, by Ascending Order of DMFT", from the Disease Control Priorities Project. Page accessed January 8, 2007.
- ↑ 92.0 92.1 92.2 92.3 92.4 Epidemiology of Dental Disease, hosted on the University of Illinois at Chicago website. Page accessed January 9, 2007.
- ↑ 93.0 93.1 93.2 93.3 93.4 93.5 93.6 Suddick RP, Harris NO (1990). "Historical perspectives of oral biology: a series". Crit Rev Oral Biol Med. 1 (2): 135–51. PMID 2129621. http://crobm.iadrjournals.org/cgi/reprint/1/2/135.pdf.
- ↑ Richards MP (December 2002). "A brief review of the archaeological evidence for Palaeolithic and Neolithic subsistence". Eur J Clin Nutr 56 (12): 16 p following 1262. doi:10.1038/sj.ejcn.1601646. PMID 12494313. http://www.nature.com/ejcn/journal/v56/n12/full/1601646a.html.
- ↑ 95.0 95.1 History of Dentistry: Ancient Origins, hosted on the American Dental Association website. Page accessed January 9, 2007.
- ↑ Dig uncovers ancient roots of dentistry: Tooth drilling goes back 9,000 years in Pakistan, scientists say, hosted on the MSNBC website . Page accessed January 10, 2007.
- ↑ Anderson T (October 2004). "Dental treatment in Medieval England". Br Dent J 197 (7): 419–25. doi:10.1038/sj.bdj.4811723. PMID 15475905. http://www.nature.com/bdj/journal/v197/n7/full/4811723a.html.
- ↑ Elliott, Jane. Medieval teeth 'better than Baldrick's', hosted on the BBC news website. October 8, 2004. Page accessed January 11, 2007.
- ↑ Salma Almahdi (2003), "Muslim Scholar Contribution in Restorative Dentistry", Journal of the International Society for the History of Islamic Medicine 2, pp. 56-57.
- ↑ Gerabek WE (March 1999). "The tooth-worm: historical aspects of a popular medical belief". Clin Oral Investig 3 (1): 1–6. doi:10.1007/s007840050070. PMID 10522185. http://link.springer.de/link/service/journals/00784/bibs/9003001/90030001.htm.
- ↑ McCauley, H. Berton. Pierre Fauchard (1678-1761), hosted on the Pierre Fauchard Academy website. The excerpt comes from a speech given at a Maryland PFA Meeting on March 13, 2001. Page accessed January 17, 2007.
- ↑ Kleinberg I (1 March 2002). "A mixed-bacteria ecological approach to understanding the role of the oral bacteria in dental caries causation: an alternative to Streptococcus mutans and the specific-plaque hypothesis". Crit Rev Oral Biol Med. 13 (2): 108–25. doi:10.1177/154411130201300202. PMID 12097354. http://crobm.iadrjournals.org/cgi/pmidlookup?view=long&pmid=12097354.
- ↑ Baehni PC, Guggenheim B (1996). "Potential of diagnostic microbiology for treatment and prognosis of dental caries and periodontal diseases". Crit Rev Oral Biol Med. 7 (3): 259–77. doi:10.1177/10454411960070030401. PMID 8909881. http://crobm.iadrjournals.org/cgi/reprint/7/3/259.pdf.
References
|
Printed sources
|
Online sources
|
External links
- What causes cavities; an indepth look
- Links to tooth decay pictures (Hardin MD/Univ of Iowa)
- Diet, Nutrition and the prevention of chronic diseases (including dental caries) by a Joint WHO/FAO Expert consultation (2003) .
- Global Oral Health - CaPP, a chart containing caries data from selected countries.
- Common questions on dental caries treatment
- Sugar, acid and teeth
|
||||||||||||||||||||||||||